Reading time: 04 minutes
Team PSP | 08/02/2024
In the rapidly evolving landscape of 2024, the fight against cancer has moved beyond the “blunt instruments” of the past. While chemotherapy and radiation remain staples, we have entered the era of the Precision Immune Strike. At the heart of this shift is Immunotherapy, specifically the move from the widely known CAR-T therapies to the next-frontier: TCR-T Cell Therapy.
As we look at the clinical successes of this year, it’s clear that we are no longer just treating cancer; we are reprogramming the body to outsmart it.
Understanding the Immune Breakthrough
The immune system’s natural ability to hunt abnormal cells is evidenced by Tumor-Infiltrating Lymphocytes (TILs). However, cancer cells are masters of disguise, using surface proteins to “turn off” immune responses. Immunotherapy acts as the ultimate software update for your white blood cells, stripping away the cancer’s invisibility cloak.
TCR-T vs. CAR-T: A 2024 Comparison
While CAR-T (Chimeric Antigen Receptor) therapy has revolutionized the treatment of blood cancers (leukemias and lymphomas), TCR-T (T-Cell Receptor) therapy is the 2024 “game-changer” for solid tumors.
Feature | CAR-T Cell Therapy | TCR-T Cell Therapy |
Target Location | Surface-level proteins only. | Surface and intracellular proteins. |
Antigen Scope | Limited to “man-made” targets. | Broad, utilizing natural T-cell receptors. |
Solid Tumor Efficacy | Challenged by “off-target” toxicity. | Superior at identifying solid tumor markers. |
Mechanism | Synthetic receptors. | Genetically optimized natural receptors. |
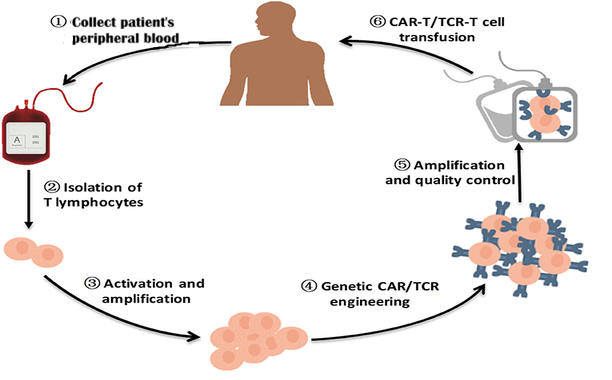
How TCR-T Therapy Works: The Five-Step Process
- Leukapheresis: T-cells are harvested from the patient’s blood.
- Genetic Reprogramming: Using CRISPR-Cas9 (the gold standard in 2024), scientists insert a specific gene into the T-cells so they can express a high-affinity T-cell receptor.
- Expansion: The “Super T-cells” are grown by the millions in a bioreactor.
- Lymphodepletion: The patient receives mild chemotherapy to clear space in the immune system.
- Infusion: The modified cells are returned to the patient, where they hunt for specific HLA (Human Leukocyte Antigen) markers.
Why TCR-T is Dominating the 2024 Pipeline
- Intracellular Access: Most cancer-specific proteins live inside the cell. CAR-T can’t see them. TCR-T can “read” the fragments of these internal proteins displayed on the cell surface, allowing it to target lung, breast, prostate, and colon cancers.
- Reduced “Antigen Escape”: By targeting multiple parts of a single cancer protein, TCR-T makes it much harder for a tumor to mutate and hide.
- Predictable Safety: Because TCRs rely on natural MHC (Major Histocompatibility Complex) recognition, the risk of the “cytokine storms” seen in early CAR-T trials is more manageable in 2024 protocols.
Challenges on the Horizon
Despite the robust growth—with the CAR-T market alone valued at $2.87 billion in 2023 and growing—hurdles remain:
- Manufacturing Costs: Creating a personalized “living drug” for every patient is resource-intensive.
- HLA Restriction: Currently, most TCR-T therapies are “locked” to specific HLA types (like HLA-A*02.01). The 2024 goal is to expand these libraries to cover a more diverse global population.
The Future: Personalized Immunotherapy
The convergence of AI-driven antigen discovery and CRISPR-Cas9 has turned a “one-off” experimental treatment into a scalable therapeutic platform. As research progresses through the end of the year, TCR-T therapy is moving from a last-resort option for refractory tumors to an earlier-stage intervention.
In 2024, we aren’t just fighting cancer; we are engineering its obsolescence.
