Reading time: 05 minutes
Team PSP | 17/03/2022
As we move through 2022, the landscape of cancer treatment is undergoing a seismic shift. In 2020 alone, cancer claimed nearly 10 million lives globally. While conventional weapons like chemotherapy, radiation, and surgery have been our primary defense for decades, their “one-size-fits-all” approach often struggles against the complex heterogeneity of cancer cells.
Enter Chimeric Antigen Receptor-T cell (CAR-T) therapy. No longer just a concept of the future, CAR-T has emerged as one of the most promising “living drugs” in our medical arsenal. By genetically reprogramming a patient’s own immune system to identify and incinerate malignant cells, we are seeing durable clinical responses that were once thought impossible.
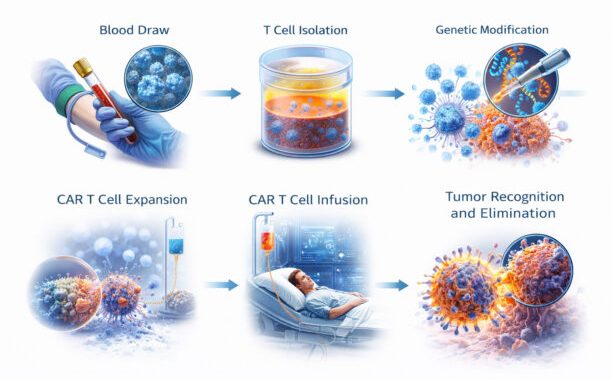
How CAR T-Cell Therapy Works: Engineering an Internal Army
CAR-T is a form of immunotherapy that turns T-cells—the “soldiers” of our immune system—into precision-guided missiles.
- Recognition: Scientists engineer T-cells with Chimeric Antigen Receptors (CARs). These are synthetic surface receptors designed to recognize specific proteins (antigens) found on the surface of cancer cells.
- Binding: Once infused back into the patient, these engineered cells seek out tumor cells. The extracellular domain of the CAR binds to the tumor antigen like a key into a lock.
- Activation: This binding triggers the intracellular signaling domain, activating the T-cell to launch a cytotoxic attack, destroying the cancer cell.
- Persistence: Unlike traditional drugs that are metabolized and exit the body, CAR T-cells multiply into hundreds of millions and can live in the body for months, providing ongoing surveillance against relapse.
The Manufacturing Journey: From Bench to Bedside
Manufacturing these “living drugs” is a highly complex, customized process that requires elite coordination:
- Leukapheresis: The process begins by drawing the patient’s blood through a machine to isolate white blood cells (lymphocytes). The remaining blood components are returned to the patient.
- Genetic Modification: In specialized labs, viral vectors are used to “infect” the T-cells with the CAR gene, permanently modifying their genome to express the new receptors.
- Expansion in Bioreactors: Since millions of cells are needed, these engineered T-cells are grown in a bioreactor until they reach a therapeutic volume.
- Pre-conditioning: Before the “army” is sent back in, the patient undergoes mild chemotherapy to deplete existing lymphocytes. This clears the way and reduces “competition” for the new CAR T-cells.
2022: A Growing Investigative Pipeline
The medical community is currently tracking over 500 CAR T-cell therapies in clinical trials. While early success was found in hematologic malignancies (blood cancers) using the CD-19 biomarker, the pipeline is expanding rapidly:
|
Product |
Manufacturer |
Approved Use (as of 2022) |
|
Yescarta |
Kite/Gilead |
Large B-cell lymphoma (First FDA approval in this category). |
|
Kymriah |
Novartis |
ALL in children and young adults; B-cell lymphoma. |
|
Tecartus |
Kite/Gilead |
Mantle cell lymphoma (MCL) and adult ALL. |
|
Breyanzi |
Bristol Myers Squibb |
Relapsed or refractory large B-cell lymphoma. |
Latest Breakthroughs: In late 2021, Novartis unveiled its T-Charge™ platform. This next-gen technology allows CAR T-cells to expand primarily inside the patient’s body rather than in a lab for weeks. This could revolutionize the implementation process, making it faster and more efficient.
Future Frontiers: Overcoming Challenges
Despite its 90% remission rates in some trials, CAR-T still faces hurdles that researchers are tackling head-on this year:
- Solid Tumors: Most current therapies target blood cancers. Trials are now aggressively pursuing targets like HER2, Meso, and EGFRvIII to take the fight to solid tumors like breast and lung cancer.
- Safety (CRS): Cytokine Release Syndrome (CRS) remains a serious side effect. However, new prophylactic interventions and biomarker monitoring are making the treatment significantly safer in 2022.
- Allogeneic “Off-the-Shelf” Therapy: Currently, most therapies are autologous (using the patient’s own cells). The next frontier is allogeneic therapy—using healthy donor cells to create “off-the-shelf” treatments that don’t require the weeks-long manufacturing wait.
Conclusion
The paradigm of oncology has officially shifted. CAR T-cell therapy has moved from a “last-resort” experimental treatment to a cornerstone of modern cancer care. As we expand the scope of target molecules and streamline manufacturing through platforms like T-Charge, we are moving closer to a world where “incurable” is a term of the past.
